Details
AgilePulse Needle Array Electrodes are miniature needles designed to provide superior, highly-uniform electric fields in dermal or muscular tissue as part of the AgilePulse in vivo Gene Delivery Electroporation System. Applications for these electrodes include intra-dermal DNA vaccine research and antibody production, cancer chemotherapy research or intra-muscular gene therapy. The miniature parallel-needle array is inserted directly into the target site for fast, reliable in vivo electroporation. The electric fields produced are the closest approximation of parallel plate electrodes, treating approximately 80% of the target area with 95% of the applied electric field.
The needle array requires the needle array handle which includes the electrode connector cable. Several needle lengths and row spacing are available for various intra-dermal or intra-muscular applications. Needle Arrays can be used for up to 500 pulses with proper care and maintenance.
FEATURES
- Uniform, reliable electric fields
- Miniature needles minimize tissue trauma
- Disposable, sterile
- Medical-grade plastic and surgical steel construction
- Safety-assured design
- Multiple configurations and sizes to choose
Use of the AgilePulse HT system (formerly Cyto LVT-P) is subject to an annual license agreement between Cellectis bioresearch and the purchaser. Please contact Cellectis bioresearch cytopulse@cellectis.com for further details.
Use of the AgilePulse In Vivo system (formerly the DermaVax device) is not authorized for any veterinary and human therapeutic uses. Contact Cellectis therapeutics contact@cellectis-therapeutics.com for such uses.
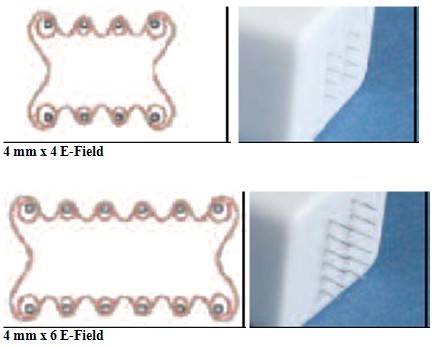
INTRA DERMAL NEEDLE ARRAYS
| Item # | 47-0040 | 47-0043 | 47-0045 | 47-0050 | 47-0060 |
| Needle Material | Stainless Steel | Stainless Steel | Stainless Steel | Stainless Steel | Stainless Steel |
| Needle Tip | Fine Point | Fine Point | Fine Point | Fine Point | Fine Point |
| Spacing in Row | 1.5 mm | 1.5 mm | 1.5 mm | 1.5 mm | 1.5 mm |
| Spacing Between Rows | 4 mm | 4 mm | 4 mm | 4 mm | 4 mm |
| Needles per Row | 4 | 3 | 4 | 6 | 6 |
| Needle Diameter | 0.3 mm | 0.3 mm | 0.3 mm | 0.3 mm | 0.3 mm |
| Needle Length | 2 mm | 3 mm | 5 mm | 2 mm | 2 mm |
INTRA MUSCULAR NEEDLE ARRAYS
| Item # | 47-0070 | 47-0080 | 47-0086 |
| Needle Material | Stainless Steel | Stainless Steel | Stainless Steel |
| Needle Tip | Fine Point | Trocar Point | Trocar Point |
| Spacing in Row | 1.5 mm | 1.5 mm | 1.5 mm |
| Spacing Between Rows | 6 mm | 6 mm | 6 mm |
| Needles per Row | 6 | 6 | 6 |
| Needle Diameter | 0.3 mm | 0.7 mm | 0.7 mm |
| Needle Length | 10 mm | 25 mm | 16 mm |
- DNA vaccine delivery
- Cancer vaccine research
- Intra-dermal transfection
- Gene therapy research
- Intra-muscular gene delivery
- Chemotherapeutic research
AgilePulse Electrodes Technical Specifications
REFERENCES
Lundberg K, Roos A-K, Pavlenko M, Wehrum D, Pisa P. Peptide specificity of HLA-A2-restricted CD8+ T cell responses induced with DNA vaccines coding for human and rhesus PSA. Vaccine. 2009 Mar 4;27(10):1557-65. Epub 2009. Jan 24 Br
Bråve A, Hallengärd D, Gudmundsdotter L, Stout R, Walters R, Wahren B, Hallermalm K. Late administration of plasmid DNA by intradermal electroporation efficiently boosts DNA-primed T and B cell responses to carcinoembryonic antigen. Vaccine. 2009 Jun 8;27(28):3692-6. Epub 2009 May 3.
Roos, A-K, Eriksson, F, Walters, D, Pisa, P, King, A. Optimization of skin electroporation in mice to increase tolerability of DNA vaccine delivery to patients. Molecular Therapy, 2009 Sep;17(9):1637-42. Epub 2009 Jun 16.
Lladser A, Ljungberg K, Tufvesson H, Tazzari M, Roos A, Quest FG, Kiessling R. Intradermal electroporation with a survivin DNA vaccine induces CTLs against a self-epitope, suppresses angiogenesis and confers long-term protection against mouse.



 800-272-2775
800-272-2775
